Could a probiotic help strengthen the gut in ACLD?
01 March 2026
In advanced chronic liver disease (ACLD), the damage isn’t confined to the liver itself. Deep in the gut, a quiet breakdown is often underway.
As disease progresses, the balance of bacteria in the gut shifts. The protective lining of the intestine becomes weaker, inflammation increases, and together, these changes can drive complications that lead to hospital admission and, in the most serious cases, organ failure.
Now, new research led by the ACLD Experimental & Therapeutic Research Group at the Roger Williams Institute of Liver Studies suggests targeting the gut microbiome may offer a promising new approach.
Published today in The International Journal of Pharmaceutics, the study explores how a multi-strain probiotic could help influence the gut environment in advanced liver disease.
Strengthening the body's essential filter
The liver and the gut are closely connected. When the gut barrier is strong, it acts as a filter, preventing harmful bacteria and toxins from entering the bloodstream. But in ACLD, that barrier can become ‘leaky’.
At the same time, the mix of bacteria in the gut changes in ways that promote inflammation. This combination of microbial imbalance and immune activation is thought to play a key role in disease progression.
That’s why researchers are increasingly asking the same important question: if we can restore balance in the gut, could we help protect ACLD patients from further harm?

Symprove, the multi-strain probiotic used in the study.
Testing a probiotic in a lifelike gut model
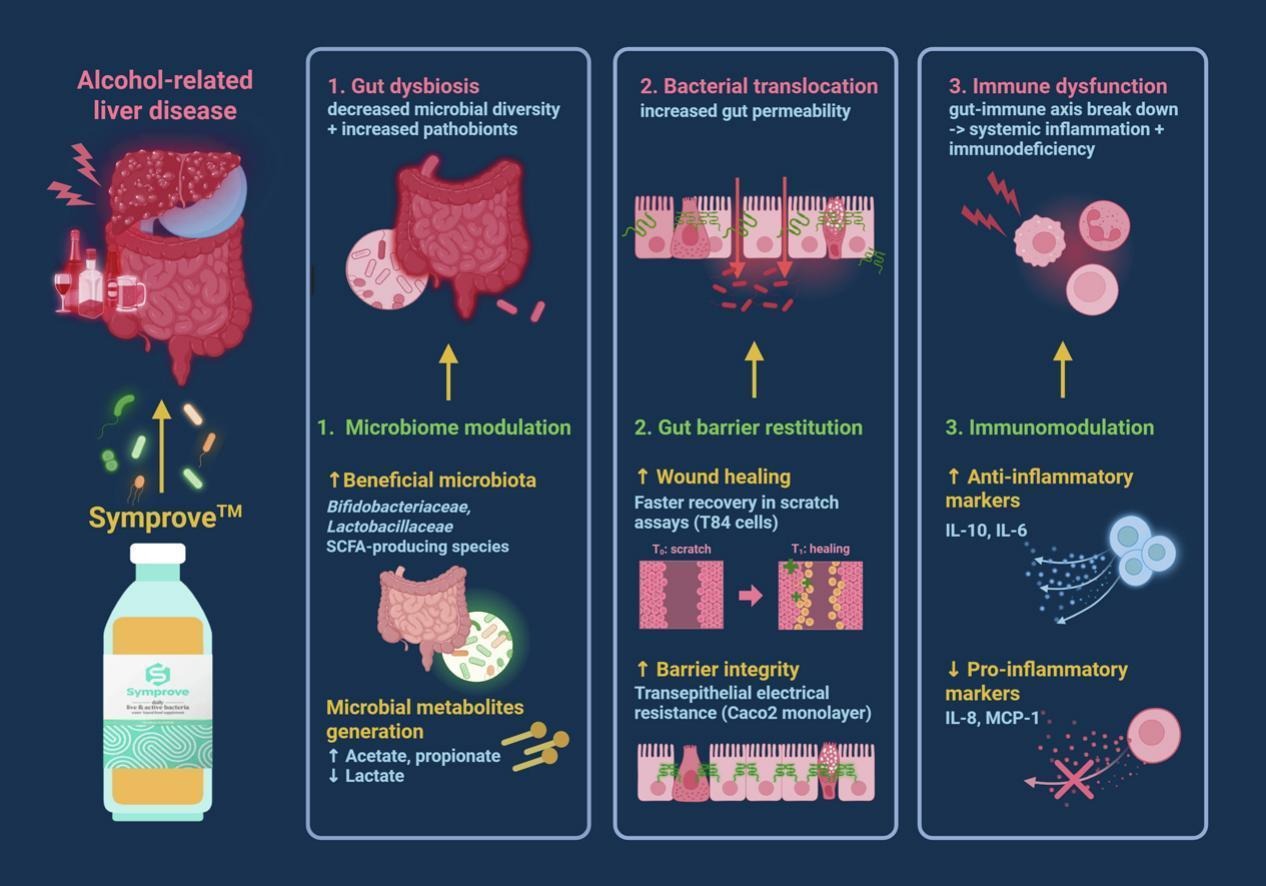
To explore this, the ACLD research group, in collaboration with industry partners through an investigator-initiated study, used an advanced lab model that recreates key features of the human gut. They introduced faecal samples from people with alcohol-related ACLD, then treated the system with a live, multi-strain probiotic preparation.
Over 48 hours, they closely monitored what happened and found encouraging results.
The probiotic altered the composition of gut bacteria in a positive way. Markers of gut barrier strength improved, suggesting tighter junctions between cells and better protection against leakage. And the team also observed faster wound healing in intestinal cells exposed to the probiotic.
There were also favourable changes in immune signalling. Anti-inflammatory molecules increased, while several pro-inflammatory mediators fell. Production of beneficial short-chain fatty acids (compounds known to support gut health) also improved.
Taken together, the findings point to a shift towards a healthier, more resilient gut environment.
From lab insight to clinical trials
The work was carried out in a sophisticated laboratory model rather than in patients themselves, involving samples from a small number of donors. This means the results aren’t ready to be translated directly into clinical practice.
However, the mechanistic data is strong. It gives us a clear scientific rationale for testing this probiotic approach in people with ACLD through carefully-designed clinical trials.
For a condition where treatment options are limited and complications can escalate quickly, that’s vital progress.
Why does this matter?
Infections and inflammation are major drivers of poor outcomes in advanced cirrhosis. Treatments that can strengthen the gut barrier, restore healthier microbial function, and reduce harmful inflammatory signalling could help reduce complications – which is especially important in a condition where options are limited.
The study also reflects a broader shift in liver research. Instead of focusing only on the damaged liver itself, scientists are increasingly looking at the whole gut-liver axis and how different organs interact.

“In advanced cirrhosis, the community of bacteria in the gut becomes seriously unbalanced, and the gut lining can no longer protect the body as it should. In our laboratory model, the probiotic helped restore some of that balance, strengthened the gut barrier and supported repair of the intestinal lining. These findings give us a foundation for future studies in people living with cirrhosis.”
– Dr Maria-Emanuela Maxan, Hepatology Clinical Research Fellow and PhD candidate at The Roger Williams Institute of Liver Studies

“People living with advanced cirrhosis urgently need new ways to reduce their risk of (and ideally prevent) infections, especially approaches that do not rely on antibiotics, which can inadvertently contribute to developing antimicrobial resistance. Our results suggest that targeting the dysfunctional gut microbiome could be part of that solution. The next step is to run carefully-designed clinical trials to understand who might benefit and how best to use this strategy in practice.”
– Dr Vishal Patel, Group Lead and Adjunct Reader in Hepatology at The Roger Williams Institute of Liver Studies, King's College London and a clinician scientist based at King's College Hospital, who led this study.
By combining expertise in microbiology, immunology and advanced in vitro modelling, and together with our academic and industry partners, the Roger Williams Institute of Liver Studies continues to lead the way in translating complex science into real treatment possibilities.
If future trials confirm the study’s findings, modulating the gut microbiome could one day become an integral part of managing ACLD.
Click here to read the paper in full. Abstract below:
Read other news articles