The “Liver Metabolism and NAFLD” group specialises in a truly-fashioned translational research strategy (“from bedside to bench and vice-versa”) that implies system biology approaches to identify key dysregulated processes, combined with translational validation (in vitro, in vivo) of potential targets/mechanisms linked to MetS/liver disease.
Projects:
- Dissecting the mechanisms through which Adipose Tissue (AT) dysfunction and systemic metabolic stress dysregulate hepatic/lipoprotein metabolism thus promoting/worsening fatty liver disease and cardio-metabolic risk;
- Identifying lipid-related metabolic pathways leading to hepatic damage (lipotoxicity) and dysregulation in wound healing processes: implications in chronic liver disease and liver regeneration;
- Dissecting the metabolic control of cell proliferation to better understand the relationship between Obesity, Metabolic Syndrome and Liver Cancer (Hepatocellular carcinoma, HCC).
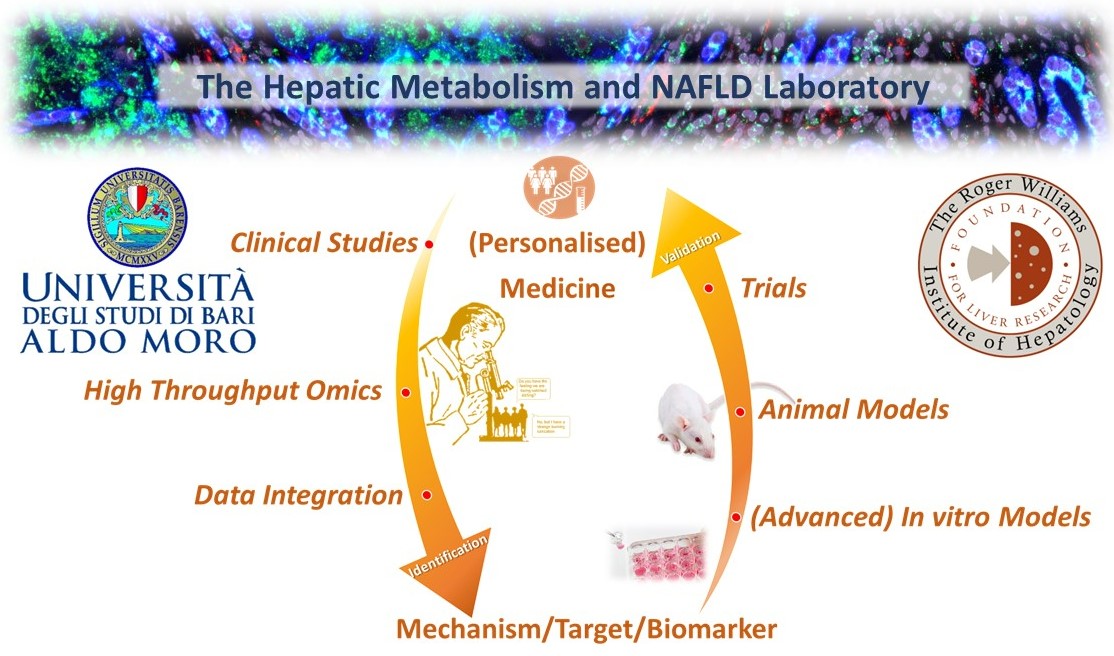
Key collaborations within the Institute:
- Shilpa Chokshi (Topics: Immuno-metabolism, PCLS)
- Luca Urbani (Topics: ECM and hepatocyte function)
- Vishal Patel (Topics: System Biology in human chronic liver disease)
External Collaborations:
NASH/HCC Pathophysiology, Liver Fibrosis. [Human Cohorts] Mike Allison (NASH - Cambridge, UK); Quentin Anstee (Newcastle, UK); EPoS & Litmus Collaborators (European NAFLD Registry); [Murine studies] Fiona Oakley & Derek Mann (Fibrosis - Newcastle, UK), Detlef Shuppan (Fibrosis - Mainz, Germany), Christian Trautwein (Inflammation - RWTH Aachen, Germany);[Advanced Culturing Systems] Luca Urbani & Shilpa Chokshi (RW-IoH, UK); Tomasz Kostrzewski (CN Bio Innovations, Cambridge, UK); Massimo Pinzani & Krista Rombouts (London UCL, UK); [Stem Cells, HCC Organoids, WNT signalling] Meritxell Huch (MPI-CBG, Germany); [TGFβ/BMP signalling] Wei Li (Cambridge, UK); Lopa Mishra (NYC, USA); [Nuclear Receptors & Coactivators; Human obesity/MetS cohorts] Antonio Moschetta (Bari, Italy).
NASH/HCC Large Omics & Data Analysis. [Metabolomics] Jules Griffin (Aberdeen, UK), Zoe Hall (London, UK), Albert Koulman (Cambridge, UK); [Proteomics] Richard Kay (Cambridge, UK); [Bioinformatics] Evangelia Petsalaki (EMBL-EBI, UK), Davide Chiarugi (Cambridge, UK).
Obesity, Systemic Metabolism, metabolic phenotyping. [Murine studies] Antonio Vidal-Puig, Sam Virtue (Cambridge, UK); [Human Cohorts] Mattia Frontini (Functional Genomics - Exeter, UK); Mario Siervo (Nitrates, intermediate metabolism, stable isotopes - Nottingham, UK); [Nutrition & Polyphenols] Daniele Del Rio (Parma, Italy) and Sumantra (Shumone) Ray (Cambridge, UK).
Team:
Dr Michele Vacca, Group Leader
Dr Gabriele Mocciaro, BSc, MSc, PhD.
Dr Arturo Simoni Nieves, BSc, MSc, PhD
Elena Maria Grao Cruces (Visiting PhD Student, Universidad de Sevilla, Spain)
Key publications:
RELEVANT ORIGINAL ARTICLES
1. Mocciaro G, Allison M, Jenkins B, Azzu V, Huang-Doran I, Herrera-Marcos LV, Hall Z, Murgia A, Susan D, Frontini M, Vidal-Puig A, Koulman A*, Griffin JL*, Vacca M*. Non-alcoholic fatty liver disease is characterised by a reduced polyunsaturated fatty acid transport via free fatty acids and high-density lipoproteins (HDL). Mol Metab. 2023 Apr 19:101728. doi: 10.1016/j.molmet.2023.101728. (IF2021: 8.568) - PMID: 37084865. *Corresponding Authors
2. Mocciaro G, D'Amore S, Jenkins B, Kay R, Murgia A, Herrera-Marcos LV, Neun S, Sowton AP, Hall Z, Palma-Duran SA, Palasciano G, Reimann F, Murray A, Suppressa P, Sabbà C, Moschetta A, Koulman A, Griffin JL*, Vacca M*. Lipidomic Approaches to Study HDL Metabolism in Patients with Central Obesity Diagnosed with Metabolic Syndrome Int J Mol Sci 2022 Jun 17;23(12):6786. doi: 10.3390/ijms23126786. (IF2021: 6.204) - PMID: 35743227 *Corresponding Authors
3. Hall Z, Chiarugi D, Charidemou E, Leslie J, Scott E, Pellegrinet L, Allison M, Mocciaro G, Anstee QM, Evan GI, Hoare M, Vidal-Puig A, Oakley F, Vacca M*, Griffin JL*. Lipid remodelling in hepatocyte proliferation and hepatocellular carcinoma. Hepatology 2021 Mar;73(3):1028-1044. doi: 10.1002/hep.31391 (IF2020: 17.425) - PMID: 32460431 *Last and Corresponding Authors
4. Vacca M*, Leslie J, Virtue S, Lam BYH, Govaere O, Tiniakos D, Snow S, Davies S, Petkevicius K, Tong Z, Peirce V, Juul Nielsen M, Ament Z, Li W, Kostrzewski T, Leeming DJ, Ratziu V, Allison MED, Anstee QM, Griffin JL, Oakley F, Vidal-Puig A*. Bone morphogenetic protein 8B promotes the progression of non-alcoholic steatohepatitis. Nature Metabolism 2020 Jun;2(6):514-531. doi: 10.1038/s42255-020-0214-9 (IF2020: 13.511) - PMID: 32694734 . *Corresponding Authors
5. Azzu V*, Vacca M*, Kamzolas I, Hall Z, Leslie J, Carobbio S, Virtue S, Davies SE, Lukasik A, Dale M, Bohlooly-Y M, Acharjee A, Lindén D. Bidault G, Petsalaki E, Griffin JL, Oakley F, Allison MED, Vidal-Puig A. Suppression of insulin-induced gene 1 (INSIG1) function promotes hepatic lipid remodelling and restrains NASH progression. Mol Metab 2021 Jun;48:101210. doi: 10.1016/j.molmet.2021.101210. (IF2020: 7.422) - PMID: 33722690 *Joint first Authors
6. D’Amore S, Härdfeldt J, Cariello M, Graziano G, Copetti M, Di Tullio G, Piglionica M, Scialpi N, Sabbà C, Palasciano G, Vacca M*, Moschetta A*. Identification of miR-9-5p as direct regulator of ABCA1 and HDL-driven reverse cholesterol transport in circulating CD14+ cells of patients with metabolic syndrome. Cardiovascular Research 2018 Jul 1;114(8):1154-1164. doi: 10.1093/cvr/cvy077. (IF2018: 7.014) - PMID: 29584810 * Last & Corresponding Authors
RELEVANT REVIEWS
7. Leslie J, Geh D, Elsharkawy AM, Mann DA*, Vacca M*. Metabolic dysfunction and cancer in HCV: Shared pathways and mutual interactions J Hepatol 2022 Feb 12;S0168-8278(22)00076-9. doi: 10.1016/j.jhep.2022.01.029. (IF2020: 25.083) - PMID: 35157957 * Corresponding authors
8. Azzu V, Vacca M, Virtue S, Allison M, Vidal-Puig A. Adipose tissue-liver cross talk in the control of whole-body metabolism: implications in non-alcoholic fatty liver disease. Gastroenterology 2020 May;158(7):1899-1912. doi: 10.1053/j.gastro.2019.12.054. (IF2020: 22.682) - PMID: 32061598
Full List and Metrics (SCOPUS): https://www.scopus.com/authid/detail.uri?authorId=35785284900

