The extracellular matrix of the liver is a complex network of proteins that provides support for the cells. Until recently, this has been thought of as an inactive and inert architectural buttress. Recent evidence suggests that this is instead a very active participant both of regeneration and repair, as well as disease progression and inflammation, with aberrant changes in ECM composition induced in chronic liver disease and cancer.
There is an urgent need for reliable and reproducible methods to understand how the extracellular matrix in different microenvironments influences inflammation and cancer progression as current available models do not mimic the pathogenesis of human liver injury and cancer. Bio-engineered tissues could represent a robust and biologically relevant model, where disease mechanisms and drug testing studies can be investigated.
Research:
Our research focus is the role of the extracellular matrix (ECM) in the development and progression of liver disease and cancer, which we study in novel 3D dynamic bioengineered disease models. We are interested in understanding how the cross-talk between the diseased ECM and hepatic and cancer cells can impact cell behaviors, inflammation, cancer progression and response to treatment.
We generate novel 3D bioengineered culture systems with metabolic and immunological functionality using primary cells and tissue-specific decellularized scaffolds providing a platform to study the processes underlying liver disease and spread of tumor (metastasis) and growth, and with potential for drug testing.
Team:
- Dr Sara Campinoti (Postdoctoral Researcher)
- Lai Wei (PhD student)
- Fabio Grundland Freile (PhD student)
- Tamas Sukei (PhD student)
- Bruna Almeida (Research Assistant)
- Kavitha Kirubendran (Research Assistant)
Projects:
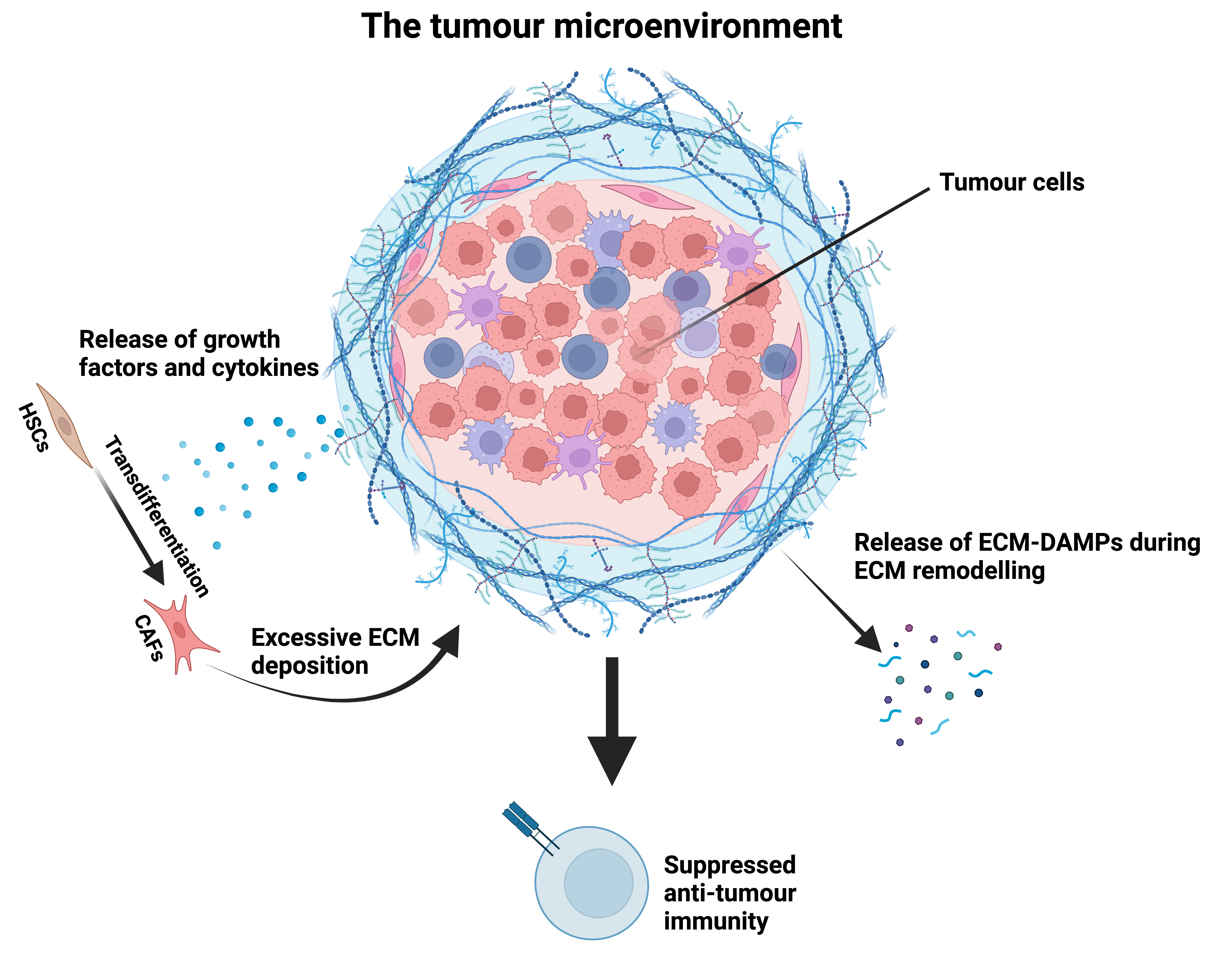
Interplay between matrix and immune cells in the context of liver cancer: we investigate the immunomodulatory properties of the remodelled extracellular matrix in primary (HCC) and secondary liver cancers. We study how matrix proteins produced by stromal cells (e.g. CAFs & HSCs) shape the tumour microenvironment and can influence anti-tumour responses.
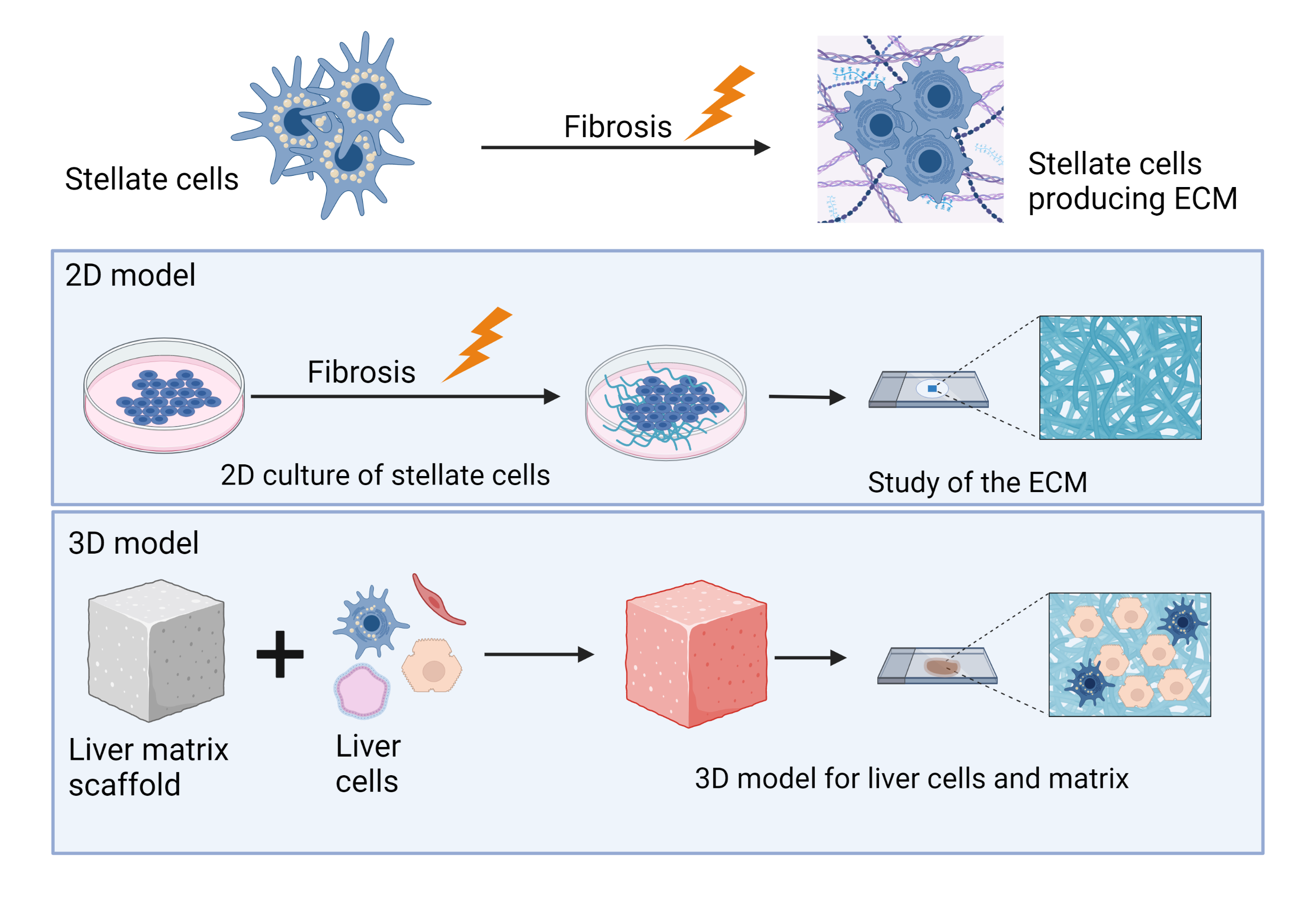
Modelling matrix deposition and turnover in liver fibrosis: study of the molecular mechanisms that drive extracellular matrix remodelling in liver fibrosis using 2D and bioengineered 3D pre-clinical in vitro models incorporating stellate cells and other liver cells. These complex models are used in anti-fibrotic drug development and testing.
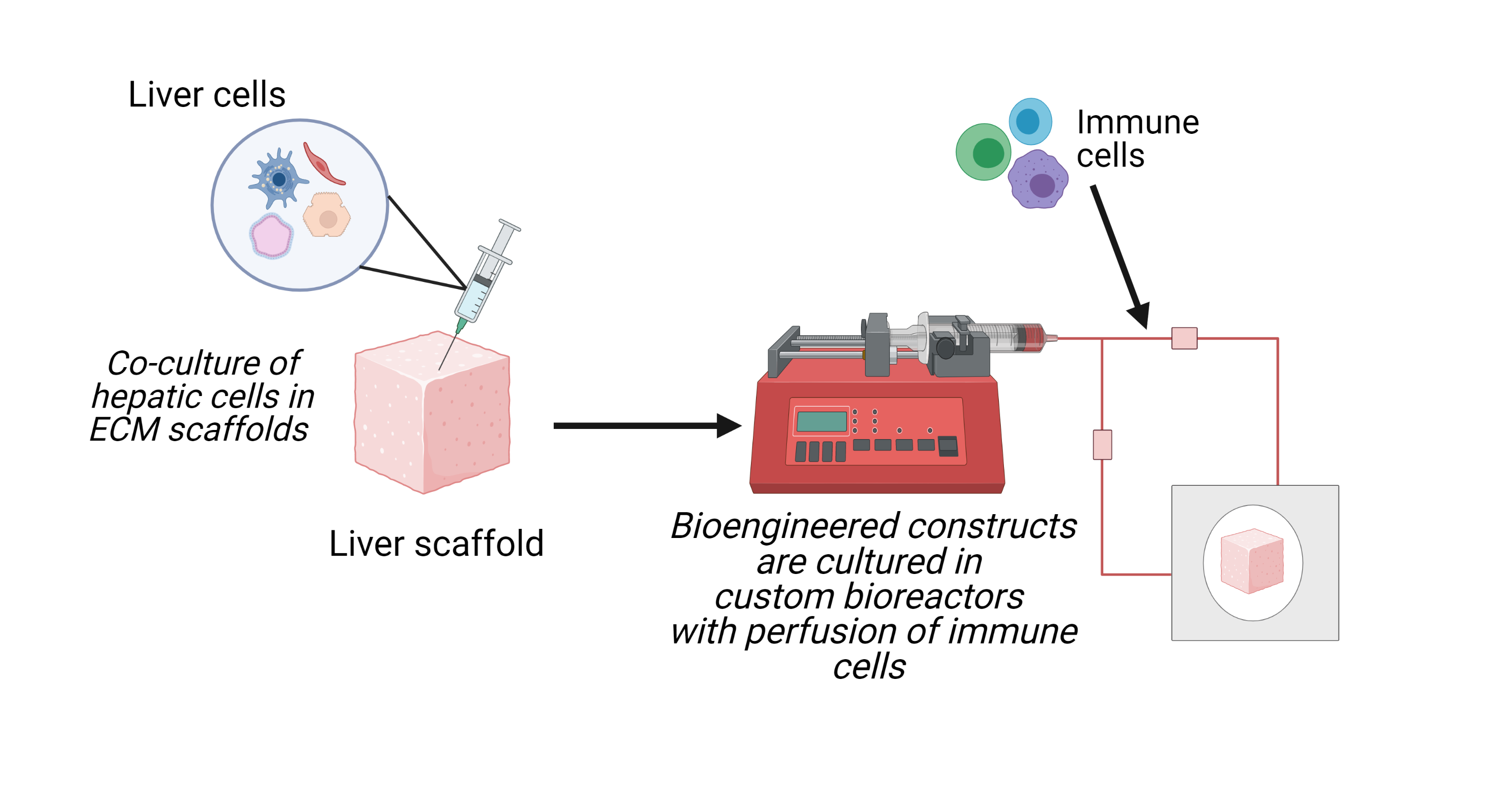
Bioengineered models of chronic liver disease: using bioengineering, we generate complex models of chronic liver disease (CLD) with human liver cells and biomaterials in bioreactor-based culture systems. These “engineered liver” models allow for the study of the crosstalk between liver microenvironment components in the development and progression of CLD.
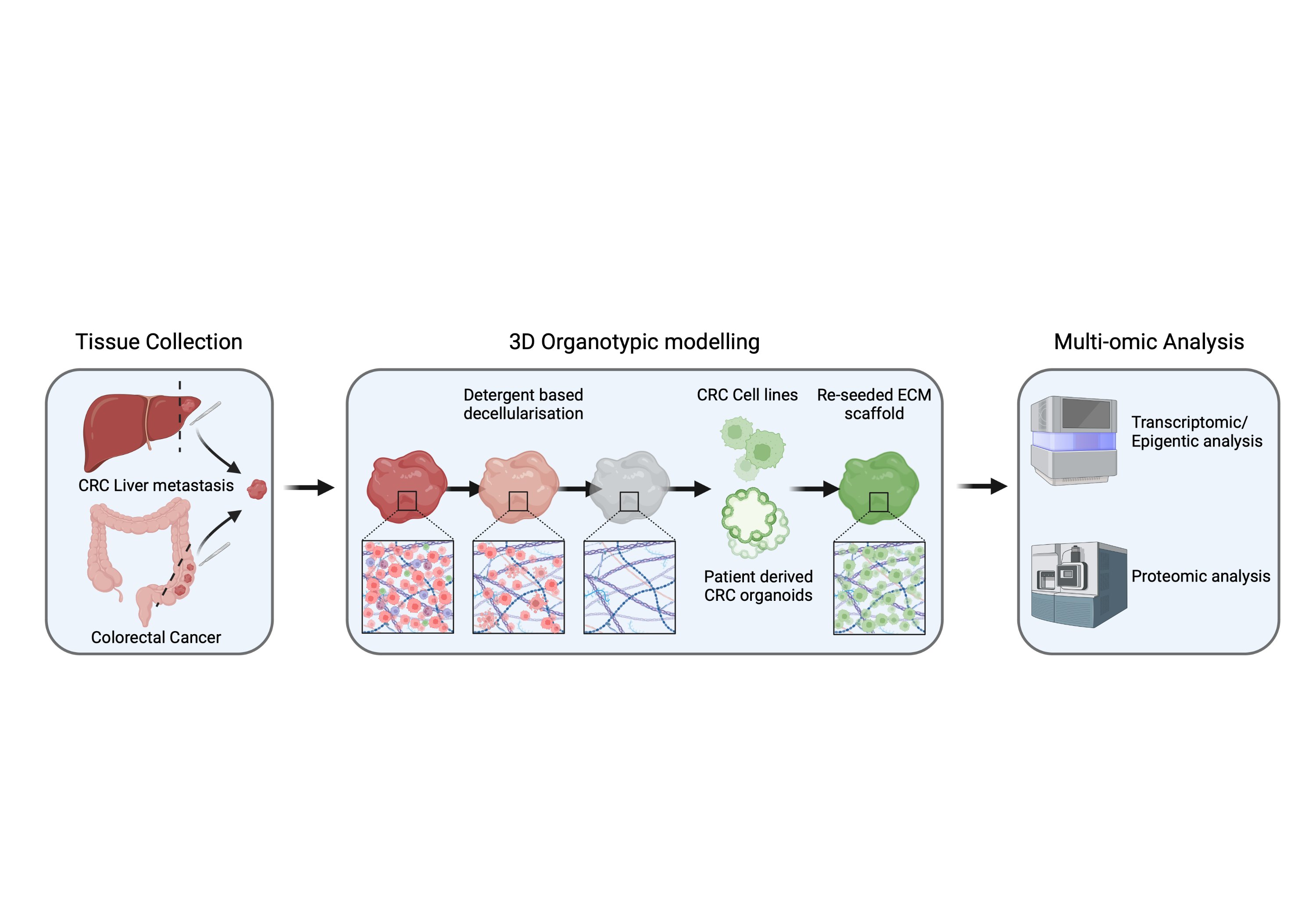
Decoding the impact of ECM signatures on tumour cell behaviour in primary and liver metastatic colorectal cancer: The project explores how changes in the matrisome of colorectal cancer (CRC) and liver metastatic sites affect cancer cell behaviour in various molecular subtypes. Patient-derived CRC/colon organoids seeded onto ECM scaffolds will be analysed using multi-omics to identify subtype-specific ECM signatures and their impact on intracellular pathways.
Modelling fatty liver disease using organoids, hydrogels and extracellular matrix: We use human liver organoids cultured in custom 3D-hydrogels and liver matrix scaffolds to mimic organ- and fatty liver disease-specific features, to understand the correlation between changes in the microenvironment and loss of hepatocyte function during disease progression.
The niche effect: study of the cellular and extracellular microenvironment of fetal liver that promotes haematopoietic stem cell expansion: multidisciplinary project to advance stem cell therapy using complex tissue engineering models and multi-omics to study the microenvironment supporting blood stem cell expansion in the liver during fetal development.
Key Collaborations:
Dr Shilpa Chokshi: her team provides essential knowledge and technical support for the study of the tumour immune microenvironment, inflammation in chronic liver disease, and the immunomodulatory properties of the extracellular matrix, key research focus points of our projects.
Dr Elena Palma: collaboration for the recruitment of patients for the collection of liver and cancer tissue specimens, essential to our disease modelling studies. Dr Palma also coordinates the testing of anti-fibrotic strategies in ex-vivo and in vitro patient-derived models of liver disease.
International Collaborations:
Prof Sara Mantero & Dr Alessandro Pellegata (Polytechnic University of Milan): established collaboration for the design and development of custom bioreactors for the culture of 3D bioengineered models.
Prof Sandra Van Vlierberghe (Ghent University): hydrogel development for the culture of hepatic organoids, to support their differentiation into hepatocyte-like cells and studies of hepatotoxicity.
Dr Michelle Holland (KCL): epigenomics and transcriptomics of bioengineered cancer models and human cancer tissue samples.
Dr Eileen Gentleman (KCL): biomechanical properties of the extracellular matrix and in vitro and ex vivo 3D models of liver disease.
Prof Paolo De Coppi (UCL): collaboration for the culture of hepatic organoids and study of the extracellular matrix biology.

